missing translation for 'onlineSavingsMsg'
Learn More
Learn More
PEAQX tetrasodium hydrate, MedChemExpress

Description
PEAQX (NVP-AAM077) tetrasodium hydrate is a potent, selective and orally active NMDA antagonist, with IC50 values of 270 nM and 29600 nM for hNMDAR 1A/2B and hNMDAR 1A/2B, respectively.
Specifications
Specifications
| Chemical Name or Material | PEAQX tetrasodium hydrate |
| Color | Earth Yellow |
| Purity Grade Notes | Research |
| Molecular Formula | C17H15BrN3Na4O6P |
| Quantity | 50 mg |
| Synonym | NVP-AAM077 tetrasodium hydrate |
| Solubility Information | H2O : 25.5 mg/mL (45.52 mM; Need ultrasonic and warming) |
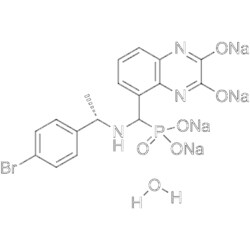
| SMILES | BrC1=CC=C([C@H](C)NC(P(O[Na])(O[Na])=O)C2=CC=CC3=C2N=C(O[Na])C(O[Na])=N3)C=C1.[H]O[H] |
| Molecular Weight (g/mol) | 560.15 |
| Formula Weight | 560.15 |
| Show More |
Research purposes only
Product Title
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.
Spot an opportunity for improvement?