
Within life sciences settings (pharmaceutical manufacturing, biotechnology, R&D) spraying a 70 percent solution of isopropyl alcohol (IPA) onto gloves prior to usage as a disinfectant is commonplace. To measure the chemical resistance of a glove material towards alcohol disinfectants, permeation tests are carried out on the gloves according to the EN 16523-1:2015 standard (determination of material resistance to permeation by chemicals: permeation by potentially hazardous liquid chemicals under conditions of continuous contact).
"In practice, when spilled on a glove, or applied on a glove for disinfection, the alcohol will evaporate rather than permeate, because in that setting it is exposed to air and not prevented from evaporating. "
Data Testing
Ansell Chemical Guardian is a database which stores permeation data tests and extrapolations for different glove materials against a range of hazardous chemicals including alcohols such as IPA and ethanol. This database test data on IPA and ethanol permeation times against four different glove materials can be seen in Table 1 and Table 2 (see below). Looking at the test data extracted, we can conclude that natural rubber latex or polyisoprene gloves have a lower resistance towards IPA and ethanol than neoprene or nitrile.
Table 1:
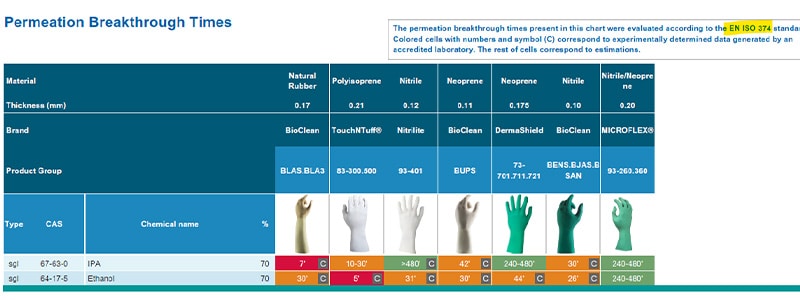
Table 2: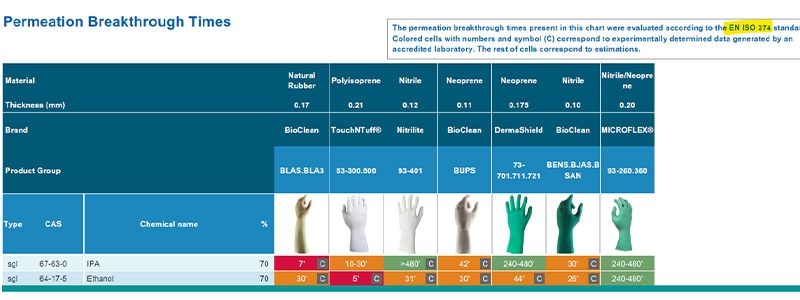
Interpretation of Theoretical Test Data for Alcohols
There can indeed be a perceived “low” resistance of clean (and sterile) gloves versus IPA, looking purely at the published permeation breakthrough times, but the following has to be noted: During the chemical permeation breakthrough test (whether this is according to standards ASTM F739, EN374-3 or EN 16523-1), the challenge chemical is completely enclosed in the test cell, with no other possibility than to permeate through the glove. Additionally, alcohols used in the test have proven to provide very variable test results.
In practice, when spilled on a glove or applied on a glove for disinfection, the alcohol will evaporate rather than permeate because in this setting it is exposed to air and not prevented from doing so. This means the lab test conditions in this case are not necessarily in line with the real application. In practice, neoprene and nitrile gloves will show good behavior towards alcohols, if the alcohol is left to evaporate naturally.
However, when there is additional rubbing together of gloved hands after alcohol treatment, it is difficult to predict how this affects the chemical resistance of the glove. It’s important to note the rubbing action could generate particles and thus add an extra contamination risk in the cleanroom.
Conclusion
If sterile gloves are purchased, there is no need to additionally disinfect gloves by applying isopropanol; however, this is often part of the standard operating procedure in cleanrooms. Although alcohols could have a deteriorative effect on the glove materials, this effect will be minimal because of the very short contact time.
Nitrile and neoprene materials will generally have better resistance to alcohols compared to natural rubber latex. We recommend against the rubbing together of gloved hands after the application of alcohol disinfectant, as this could aggravate the chemical deterioration effect, as well as generate particles into the cleanroom environment. We advise to apply the alcohol via a spray application and allow to evaporate naturally on the gloves.


